Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Collaborative Laboratories for Advanced Decommissioning Science; Osaka University*
JAEA-Review 2020-030, 55 Pages, 2020/12
The Collaborative Laboratories for Advanced Decommissioning Science (CLADS), Japan Atomic Energy Agency (JAEA), had been conducting the Nuclear Energy Science & Technology and Human Resource Development Project (hereafter referred to "the Project") in FY2019. The Project aims to contribute to solving problems in the nuclear energy field represented by the decommissioning of the Fukushima Daiichi Nuclear Power Station, Tokyo Electric Power Company Holdings, Inc. (TEPCO). For this purpose, intelligence was collected from all over the world, and basic research and human resource development were promoted by closely integrating/collaborating knowledge and experiences in various fields beyond the barrier of conventional organizations and research fields. The sponsor of the Project was moved from the Ministry of Education, Culture, Sports, Science and Technology to JAEA since the newly adopted proposals in FY2018. On this occasion, JAEA constructed a new research system where JAEA-academia collaboration is reinforced and medium-to-long term research/development and human resource development contributing to the decommissioning are stably and consecutively implemented. Among the adopted proposals in FY2018, this report summarizes the research results of the "Development of hydrogels for prevention of radioactive dust dispersion during fuel debris retrieval" conducted in FY2019. In this study, we propose coating fuel debris with thixotropic gel materials to suppress dust dispersion during debris retrieval. Hectorite clay mineral - borax composite gel was selected based on the viscosity, the transparency and the radiation resistance. Simulated cutting tests confirm that the gel coating effectively suppress the dust dispersion.
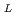 -proline methyl ester) gel membrane in LiCl solution
-proline methyl ester) gel membrane in LiCl solutionChen, J.; Asano, Masaharu; Tsubokawa, Norio*; Maekawa, Yasunari; Yamaki, Tetsuya; Yoshida, Masaru
Journal of Polymer Science, Part B; Polymer Physics, 43(20), p.2843 - 2851, 2005/10
Times Cited Count:0 Percentile:0.01(Polymer Science)Impedance spectra analysis of a thermo-responsive poly(acryloyl-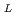 -proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by
-proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by  -radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14
-radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14 C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
Nagasawa, Naotsugu; Yagi, Toshiaki; Kume, Tamikazu; Yoshii, Fumio
Carbohydrate Polymers, 58(2), p.109 - 113, 2004/11
Times Cited Count:116 Percentile:96.14(Chemistry, Applied)A new biodegradable starch derivative hydrogel, carboxymethyl starch (CMS) hydrogel, was synthesized by irradiation in high concentrated solution (in the so-called paste-like condition). The effect of the solution concentration on the crosslinking of CMS, the properties of formed hydrogel and the biodegradability were investigated. The crosslinking of CMS was induced by irradiation at concentration range from 20 to 50 %. 1 g of the dry gel formed from the solution at concentration of 40 % crosslinked at dose of 2 kGy was able to absorb about 500 and 26 g of distilled water and 0.9 % NaCl, respectively. Biodegradation of crosslinked CMS (irradiated in 50 % solution at a dose of 5 kGy) by controlled composting was about 40 % after 2 weeks which was faster than standard cellulose powder. The contribution of crosslinking in CMS was elucidated from crosslinking behaviors of carboxymethyl amylopectin (CMAP) and carboxymethyl amylose (CMA). The crosslinked CMAP (irradiated at a concentration range from 10 to 50 %) reveal higher gel fraction and water-uptake than that of CMA. The amylopectin region in CMS is predominantly responsible for crosslinking of CMS.
Functional Materials Laboratory I
JAERI-Conf 2001-005, 273 Pages, 2001/03
no abstracts in English
L.Z.Xin*; M.Carenza*; Kaetsu, Isao*; Kumakura, Minoru*; Yoshida, Masaru; Fujimura, Takashi
Radiation Physics and Chemistry, 40(6), p.579 - 584, 1992/00
no abstracts in English
Sekine, Yurina; Chi, S.*; Fernandez Baca, J.*; Fukazawa, Tomoko*; Fukazawa, Hiroshi
no journal, ,
Hydrogels are unique polymer materials consisting of large amount of water in their three-dimensional networks. The structure of water is an important factor governing the chemical and physical properties of gel materials. To investigate structure and properties of water in poly-N,N,-dimethylacrylamide (PDMAA) gel, neutron diffraction and differential scanning calorimetry (DSC) curve of the high (90wt%) and low (55wt%) D O content gels were measured. The neutron diffraction patterns show that amorphous, cubic, and hexagonal ices exist in the low D
O content gels were measured. The neutron diffraction patterns show that amorphous, cubic, and hexagonal ices exist in the low D O content gel at low temperature, whereas only hexagonal ice exists in the high D
O content gel at low temperature, whereas only hexagonal ice exists in the high D O content gel. The DSC result shows that the low D
O content gel. The DSC result shows that the low D O content gel contains only intermediate and bound water. In contrast, high D
O content gel contains only intermediate and bound water. In contrast, high D O content gel contains free, intermediate, and bound water. Therefore the results indicate that amorphous and cubic ices are only formed when the hydrogels do not have the free water.
O content gel contains free, intermediate, and bound water. Therefore the results indicate that amorphous and cubic ices are only formed when the hydrogels do not have the free water.